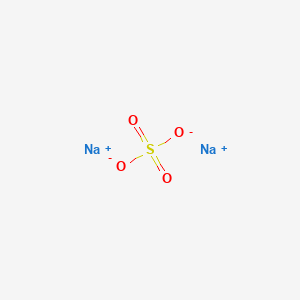


Sodium sulfate, anhydrous
CAS No.: 7757-82-6
- Molecular Formula: Na₂SO₄
- Molecular Weight: 142.04 g/mol
Chemical type
- Ionic salt
Key properties
- Provides ionic conductivity
- Used to minimize electrode reactions (compared to NaCl)
- Solubility in water at 20°C: 1.354 mol/L.
- Present in pore solution; contributes to osmotic suction.
- Can crystallize as mirabilite (Na₂SO₄·10H₂O) or thenardite (Na₂SO₄) during phase change, leading to salt expansion.
- At 0.5°C, the solubility is 50.6 g/L.
- Its addition to loess increases pore solution concentration.
- Electrolyte solution in the electrode chambers (EC) of the experimental ED setup.
- Used to prepare saline loess samples to study salt weathering mechanisms in check dams.
- Identified as the primary culprit for salt weathering hazards in field surveys of loess check dams in northern Shaanxi.
Classification by use
- Supporting electrolyte in electrochemical systems
- Chemical used in geotechnical/civil engineering research
A trustworthy factory and manufacturer
- [Cite:1] Voltage optimization strategy to reduce electric field non-uniformity and improve efficiency in electrodialysis systems, Electrochemistry Communications, Volume 183, February 2026, 108104
- [Cite:2] Evaluation of the relationship between freezing point and suction of sodium sulfate loess near 0°C, Soils and Foundations, Volume 66, Issue 2, April 2026, 101753