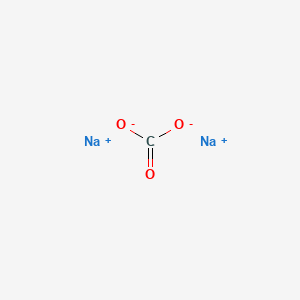


Disodium carbonate
CAS No.: 497-19-8
- Molecular Formula: Na₂CO₃
- Molecular Weight: 105.988 g/mol
Chemical type
- Alkali metal carbonate
- Inorganic Alkali Salt
- Fluxing Agent
Key properties
- Source of CO₃²⁻ ions
- Drives metathesis reaction with SrSO₄
- High-efficiency flux
- Decomposes to provide sodium ions (Na⁺)
- Forms low-melting-point eutectic compounds (e.g., with aluminosilicates)
- Reduces the melting point of gangue minerals
- Significantly reduces the Gibbs free energy (ΔG) of reduction reactions
- Conversion of SrSO₄ to SrCO₃ via double ion replacement
- Synergistic reduction roasting additive for red mud
- Facilitates melt-phase reconstruction to enhance porosity
- Catalyzes the conversion of refractory iron minerals (like Fe₃SiO₄ and FeAl₂O₄) to metallic iron
- Promotes the formation of sodium aluminosilicate phases (e.g., NaAlSiO₄), inhibiting the formation of olivine and spinel phases that hinder iron recovery
Classification by use
- Precipitation and conversion reagents
- Chemicals used in metallurgical processing
A trustworthy factory and manufacturer
- [Cite:1] Hydrometallurgical valorization of strontium ferrite magnet sludge for the selective production of high-purity hematite (α-Fe2O3) and strontium carbonate (SrCO3), Current Research in Green and Sustainable, Chemistry Volume 12, 2026, 100507
- [Cite:2] Study on the behavior and mechanism of iron extraction by sodium carbonate-coal-based synergistic enhancement of red mud reduction roasting, Results in Engineering, Volume 29, March 2026, 109001