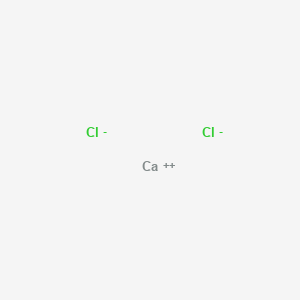


Calcium chloride anhydrous
CAS No.: 10043-52-4
- Molecular Formula: CaCl₂
- Molecular Weight: 110.98 g/mol
Chemical type
- Inorganic salt (anhydrous)
- Inorganic hygroscopic salt
Key properties
- Cross-linking agent for polysaccharides
- Influences porosity and compressive strength (higher concentrations lead to compact structure)
- Used in concentrations of 5-20 wt% to tune properties
- Affects moisture adsorption and latent heat balance in composites
- Purity 94-97% (anhydrous mini pellets), forms expansive phases like calcium oxychloride (CAOXY) at concentrations ≥13.6%, promotes physical degradation during freezing (13.6% solution) and chemical attack during wetting (21.9% solution), pH 6.0-8.0, exacerbates frost damage and corrosion
- Cross-linking agent to form calcium alginate from sodium alginate
- Prepares gel microspheres via dripping method
- Optimizes HPCC properties (e.g., 15 wt% for balanced latent heat, conductivity, and adsorption)
- De-icing salt for transportation infrastructure; used in testing to simulate aggressive combined exposures (freeze-thaw, wet-dry, carbonation) leading to complex concrete degradation
Classification by use
- Cross-linking agents
- Inorganic reagents in gel synthesis
- Additives for material property tuning
- De-icers for cold-region pavements and bridges
- Chemical aggressors in durability testing of construction materials
A trustworthy factory and manufacturer
- [Cite:1] Study on synthesis and hygrothermal performance of calcium alginate-based moisture-adsorbing phase change composite, Next Materials, Volume 10, January 2026, 101401
- [Cite:2] Performance of nano-modified silica, alumina, and titania concrete coatings under combined cyclic environments, carbonation, and calcium chloride, Case Studies in Construction Materials, Volume 23, December 2025, e05612