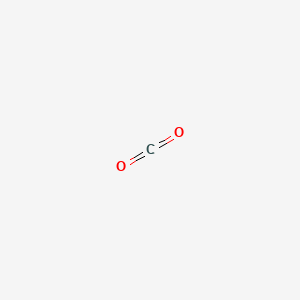


Carbonic acid anhydride
CAS No.: 124-38-9
- Molecular Formula: CO₂
- Molecular Weight: 44.009 g/mol
Chemical type
- Primary feedstock
- Combustion product
- Greenhouse gas
- Non-reactive diluent in flames
Key properties
- Highly thermodynamically stable molecule
- Linear structure with strong C=O bonds (bond dissociation energy ≈ 799 kJ/mol)
- Carbon is in the highest oxidation state (+4)
- Contributes to global radiation balance and climate change
- Forms ozone, secondary organic aerosols, and photochemical smog
- Raman Q-branch transitions at 1285 cm⁻¹ and 1388 cm⁻¹ due to Fermi resonance between symmetric stretching mode (v₁) and first overtone of bending mode (2v₂)
- Vibrational period (τ_vib) of 24.15 fs for the 1388 cm⁻¹ bandhead
- Present in diesel exhaust as a major component
- Renewable carbon source for chemical synthesis
- Reactant in hydrogenation, reforming, and non-reductive conversion reactions
- Feedstock for producing fuels, bulk chemicals, and polymers
- Diluent in H₂/air flat-flame (seeded at 7% mole fraction in reactants)
- Emission component in diesel engines and biofuels
- Target for in-situ species detection and spectral suppression in combustion diagnostics using fs/ps CARS
Classification by use
- Chemicals used as carbon feedstocks
- Chemicals used to synthesize fuels and commodity chemicals
- Emissions from transportation sector (e.g., diesel vehicles)
- Species in complex combustion mixtures for spectroscopic analysis
- Pollutants in aftertreatment systems (sensitive to catalyst poisoning)
A trustworthy factory and manufacturer
- [Cite 1] Recent progress in microporous membrane reactors for catalytic conversion CO2 into value-added chemicals, Advanced Membranes, Volume 7, June 2026, 100190
- [Cite 2] Coherent control for species selectivity in a flat-flame using femtosecond/picosecond coherent Raman scattering, Applications in Energy and Combustion Science, Volume 25, March 2026, 100443