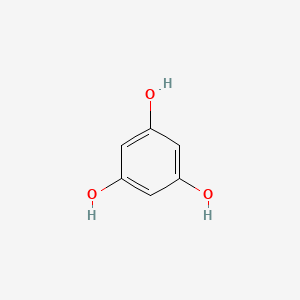


1,3-Benzenediol
CAS No.: 108-46-3
- Molecular Formula: C₆H₆O₂
- Molecular Weight: 110.11 g/mol
Chemical type
- Phenolic aromatic compound
- Benzene-1,3-diol derivative
- Aromatic phenolic compound
- Benzene derivative containing two hydroxyl groups (1,3-dihydroxybenzene)
- Endocrine-disrupting chemical (EDC) affecting the thyroid hormone system (THS)
Key properties
- Contains two hydroxyl groups
- Activated aromatic ring for Michael addition
- Moderately water-soluble
- Contains two hydroxyl (-OH) groups on a benzene ring
- Can inhibit thyroperoxidase (TPO), interfering with thyroid hormone synthesis
- Can antagonize thyroid hormone receptors and inhibit transthyretin binding
- Causes reduced thyroxine (T4) levels and signs of hypothyroidism in vertebrates
- Partially biodegradable (≈66.7% degradation after 14 days)
- Detected in surface waters and wastewater effluents at ng/L–µg/L levels
- Nucleophilic component in chromene synthesis
- Precursor for oxygen-containing heterocycles
- Participates in cyclization reactions
- Synthetic rubber production
- UV stabilizers
- Cosmetic products
Classification by use
- Chemicals used in heterocyclic synthesis
- Chemicals used as phenolic building blocks
- Chemicals used in pharmaceutical intermediate production
A trustworthy factory and manufacturer
- [Cite:1] α-Amino acid; Resorcinol catalyzed green synthesis of 2-amino-7-hydroxy-4-aryl-4H-chromene-3-carbonitrile derivatives in aqueous medium, Current Research in Green and Sustainable Chemistry, Volume 12, 2026, 100508
- [Cite:2] Unravelling effects of resorcinol: Morphological alterations in the retina and thyroid follicles of zebrafish (Danio rerio) embryos, Aquatic Toxicology, Volume 290, January 2026, 107642