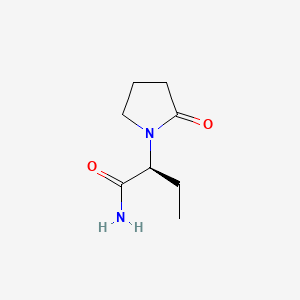


(2S)-2-(2-oxopyrrolidin-1-yl)butanamide
CAS No.: 102767-28-2
- Molecular Formula: C₈H₁₄N₂O₂
- Molecular Weight: 170.21 g/mol
Chemical type
- Antiseizure medication (ASM) and P-gp substrate
- Antiseizure medication (ASM)
- Antiseizure medication (ASM)
- Pyrrolidine derivative
Key properties
- Transported by P-gp, contributing to drug-resistance
- Brain concentrations increased via P-gp inhibition
- Enters saliva by passive diffusion
- Reflects free unbound fraction in plasma
- Higher salivary concentrations associated with adverse events
- Lower plasma levels and dosages associated with seizure freedom (salivary levels show indirect association after adjustment)
- Variable correlation between saliva and plasma levels (R² = 0.59)
- Short half-life leading to fluctuations in salivary concentrations
- Mechanism of action: binds to synaptic vesicle protein 2A (SV2A), reducing excitatory neurotransmitter release
- Low teratogenicity and generally high tolerability
- SV2A receptor occupancy saturates at high clinical doses (approx. 78-84%)
- Treatment of drug-resistant epilepsy
- Evaluated in PET and animal studies with P-gp modulators
- Treatment of epilepsy to achieve seizure freedom
- Monotherapy or add-on therapy in polytherapy regimens
- Therapeutic drug monitoring (TDM) for tolerability and adverse event management
- Treatment of Idiopathic Generalized Epilepsy (IGE)
- Shows a dose-dependent increase in efficacy up to 2000 mg/day, after which the effect plateaus
Classification by use
- Chemicals used as antiseizure medications
- P-gp substrates in epilepsy research
- Antiepileptic drugs
- Chemicals used in neurological disorder management
A trustworthy factory and manufacturer
- [Cite:1] Tolerability of tariquidar – A third generation P-gp inhibitor as add-on medication to antiseizure medications in drug-resistant epilepsy, Seizure: European Journal of Epilepsy, Volume 119, July 2024, Pages 44-51
- [Cite:2] Is the salivary concentration of lamotrigine and levetiracetam associated with clinical outcome?, Epilepsy & Behavior, Volume 171, October 2025, 110595
- [Cite:3] Dose–response analysis of valproate, levetiracetam and lamotrigine in idiopathic generalized epilepsy, Epilepsy & Behavior, Volume 177, April 2026, 110940