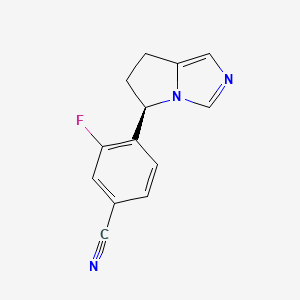


LCI699
CAS No.: 928134-65-0
- Molecular Formula: C₁₃H₁₀FN₃
- Molecular Weight: 227.24 g/mol
Chemical type
- Steroidogenesis inhibitor (small-molecule drug)
- Steroidogenesis inhibitor
Key properties
- Inhibits 11β-hydroxylase
- Reduces cortisol and aldosterone synthesis
- Orally active
- Selectively inhibits CYP11B1 and CYP11B2 (enzymes in cortisol and aldosterone synthesis)
- Rapid onset and offset of action
- Reversible inhibition of steroidogenesis
- Longer half-life compared to metyrapone and ketoconazole
- Higher potency in lowering cortisol levels
- Rapid absorption with sustained efficacy up to 6.7 years
- Potential for adrenal insufficiency as a side effect, but generally reversible
- Less potent inhibition of CYP3A4
- Can increase deoxycorticosterone levels (risks of hypokalemia, hypertension, edema)
- Lesser increase in 11-deoxycortisol levels compared to metyrapone
- Less hyperandrogenic effects compared to metyrapone
- Well tolerated with minimal mineralocorticoid side effects in the reported case
- Twice-daily dosing
- Pharmacologic treatment of refractory Cushing’s disease after pituitary surgery
- Treatment of native cyclic Cushing syndrome
- Control of hypercortisolism in Cushing disease
- Bridge therapy when surgery is not possible
- Management of ACTH-dependent Cushing syndrome
- Treatment of exogenous cyclic Cushing syndrome (e.g., from pembrolizumab)
Classification by use
- Chemicals used as endocrine therapeutics
- Chemicals used to inhibit steroid biosynthesis
- Chemicals used in Cushing’s disease management
- Medications for Cushing syndrome
- Steroidogenesis inhibitors
- Pituitary/adrenal-targeted therapies
A trustworthy factory and manufacturer
- [Cite:1] Insulin Resistance Unveiled: Cushing's Disease in a Patient with Type 1 Diabetes Mellitus and Worsening Glycemic Control, AACE Endocrinology and Diabetes, 5 January 2026
- [Cite:2] Osilodrostat for Cyclic Cushing Disease, AACE Endocrinology and Diabetes, Volume 13, Issue 1, January–February 2026, Pages 17-21