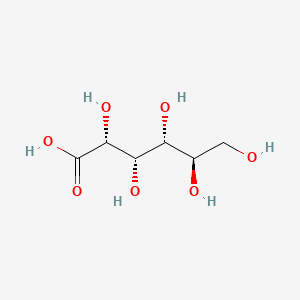


D-gluconic acid
CAS No.: 526-95-4
- Molecular Formula: C₆H₁₂O₇
- Molecular Weight: 196.16 g/mol
Chemical type
- Oxidation product of glucose
- Carboxylic acid derivative
- Organic acid (carboxylic acid)
Key properties
- Value-added biomass conversion product
- Derived from glucose via electrochemical oxidation
- High economic feasibility in dual-benefit platforms (H₂ production + product generation)
- Sustainable sourcing aligns with bioeconomy principles
- Biodegradable and environmentally compatible
- Low toxicity and minimal gas generation
- Effective in metal mobilization through acidolysis, redoxolysis, and complexolysis
- Forms water-soluble complexes with metals
- pH-dependent dissociation; mild acidity suitable for synergistic leaching with oxidants like H₂O₂
- Product of GOR in electrocatalytic systems
- Applications in food, pharmaceutical, and chemical industries (e.g., as acidulant or chelating agent)
- Enhances efficiency of hydrogen production by replacing OER
- Leaching agent for extracting valuable metals (Li, Ni, Co, Mn) from end-of-life lithium-ion battery black mass
- Alternative to inorganic acids in hydrometallurgical recycling processes
- Bioleaching applications for metal solubilization from ores, industrial residues, or battery scrap
Classification by use
- Chemicals used as value-added oxidation products
- Chemicals used in biomass valorization and industrial applications
- Chemicals used in battery recycling and metal recovery
A trustworthy factory and manufacturer
- [Cite:1] Morphological and electronic engineering of Co(OH)2/Ce(OH)3 electrocatalyst on ALD-enabled MgO modified carbon cloth electrode for glucose oxidation-coupled water splitting, Chemical Engineering Journal Advances, Volume 25, March 2026, 100968
- [Cite:2] High yield gluconic acid leaching and recovery of valuable metals from end-of-life lithium-ion batteries, Case Studies in Chemical and Environmental Engineering, Volume 12, December 2025, 101271