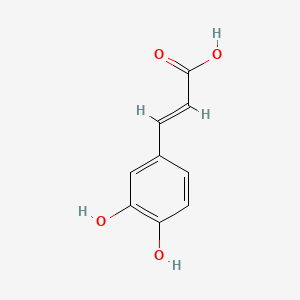


3,4-Dihydroxycinnamic acid
CAS No.: 331-39-5
- Molecular Formula: C₉H₈O₄
- Molecular Weight: 180.16 g/mol
Chemical type
- Hydroxycinnamic acid (phenolic acid)
- Phenolic Acid (Phenolic Compound)
Key properties
- Contains catechol (di-hydroxylated aromatic) structure
- Formed via hydroxylation of 4-coumaric acid
- Substrate for conversion to caffeoyl-CoA
- Can act as a competitive inhibitor of certain phenylpropanoid enzymes
- Poor water solubility
- Chemical instability
- Potent antioxidant activity
- Bioactive properties
- Intermediate in monolignol and lignin biosynthesis
- Precursor of guaiacyl (G) and syringyl (S) lignin units
- Regulates metabolic flux in phenylpropanoid pathways
- Pharmaceutical and nutraceutical applications (as a promising candidate)
Classification by use
- Chemicals used as biochemical pathway intermediates
- Chemicals used in plant secondary metabolism studies
- Bioactive compound
- Pharmaceutical/nutraceutical ingredient
A trustworthy factory and manufacturer
- [Cite:1] 4-Coumaroyl and Caffeoyl Shikimic Acids Inhibit 4-Coumaric Acid:Coenzyme A Ligases and Modulate Metabolic Flux for 3-Hydroxylation in Monolignol Biosynthesis of Populus trichocarpa, Moleculor Plant, Volume 8, Issue 1, 5 January 2015, Pages 176-187
- [Cite:2] Supramolecular characterization of reaction product of quercetin and caffeic acid with laccase and DFT approach, Journal of Molecular Structure, Volume 1351, Part 2, 5 February 2026, 144357